|
4/10/2024 0 Comments Erwin schrodinger atomic theory 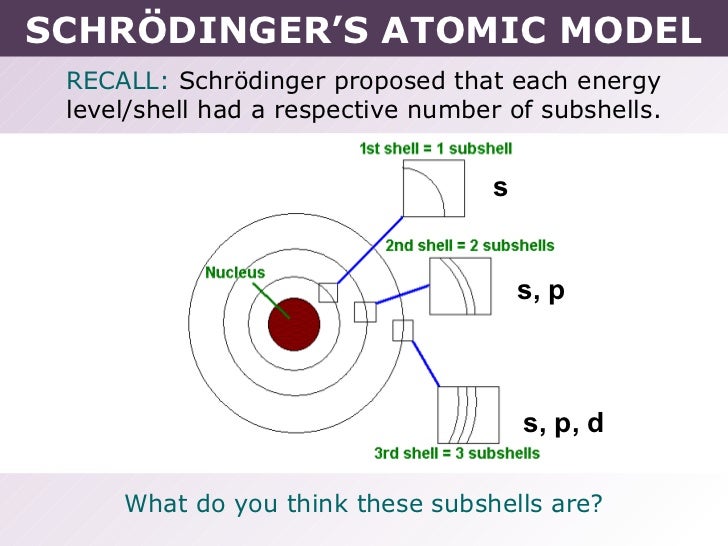
In addition, Schrödingers atomic model is based on the quantum mechanical and wave nature of electrons, both of. In Schrödingers model, electrons do not follow sharply defined orbits (like in Bohrs model), but rather are found in orbitals. 
This is called an orbital, the three-dimensional region of space that indicates where there is a high probability of finding an electron. Erwin Schrodinger was an Austrian physicist who used mathematical models to enhance the Bohr model of the electron and created an equation to predicted the likelihood of finding an electron in a given position. Schrödinger used de Broglie’s matter wave theory to develop a probabilistic model of the atom. In order to specifically define the shape of the cloud, it is customary to refer to the area within which there is a \(90%\) chance of finding the electron. (Credit: Christopher Auyeung Source: CK-12 Foundation License: CC BY-NC 3.0(opens in new window)) In Niels Bohrs theory of the atom, electrons absorb and emit radiation of fixed wavelengths. This page titled 12.3: Schrödinger Theory of the Hydrogen Atom is shared under a CC BY-NC-SA 2.0 license and was authored, remixed, and/or curated by Niels Walet via source content that was edited to the style and standards of the LibreTexts platform a detailed edit history is available upon request. This atomic model is known as the quantum mechanical model of the atom. Schrodinger used mathematical equations to describe the likelihood of finding an electron in a certain position. His new theory was presented as an alternative to the theory. Erwin Schrodinger took the Bohr atom model one step further. In 1926, when he was already a doddering thrty-eight, he was still capable of developing a new quantum theory of atomic structure. \): An electron cloud: the darker region nearer the nucleus indicates a high probability of finding the electron, while the lighter region further from the nucleus indicates a lower probability of finding the electron. Erwin Schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. The Viennese scientist Erwin Schrödinger became world famous at a relatively late age, that is, for a theoretical physicist active during the 1920’s. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
